Description
Description
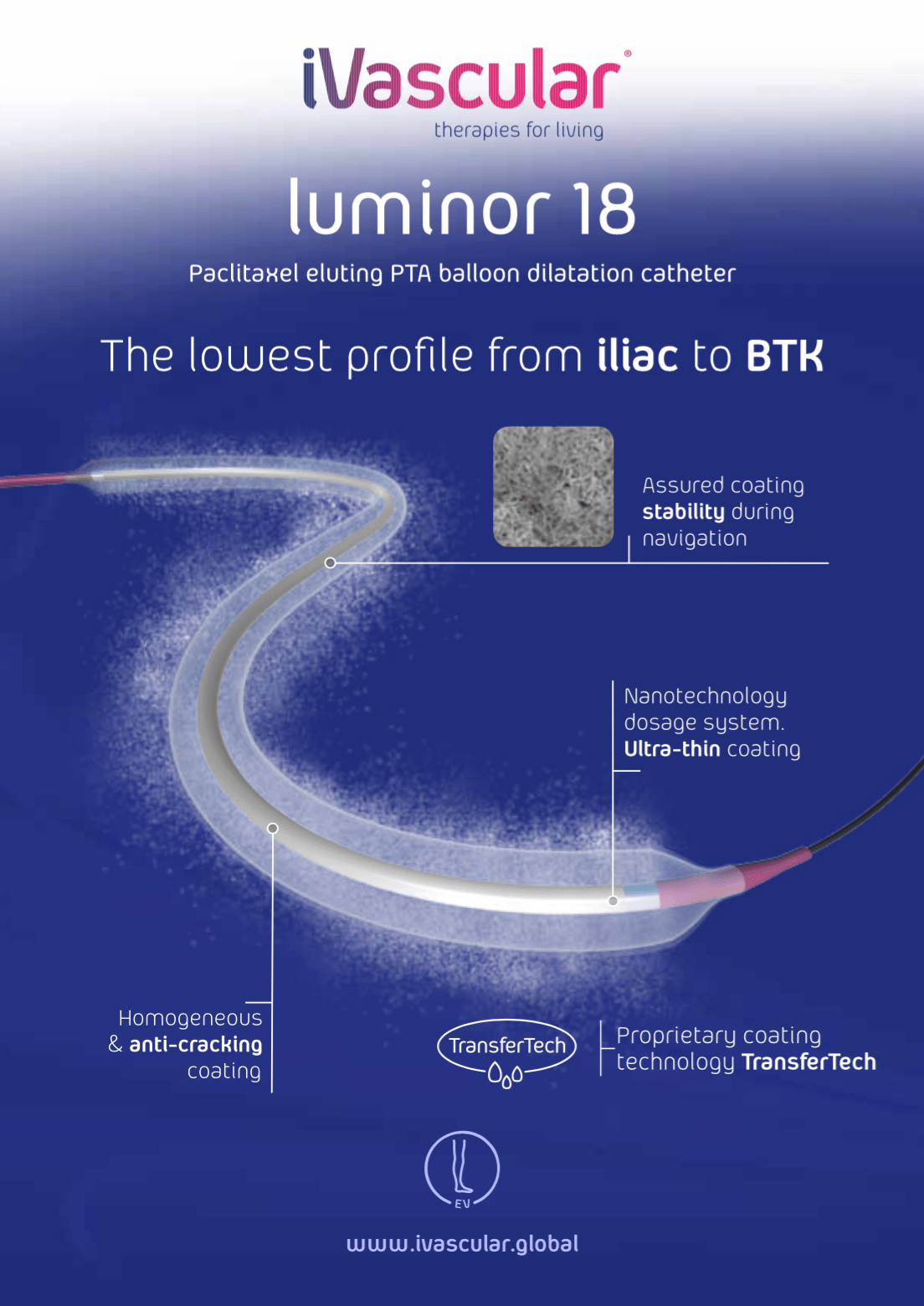
The Paclitaxel-eluting balloon “Luminor 18” is a coaxialcatheter (OTW, over-the-wire) designed for percutaneous transluminal angioplasties of small peripheral arteries.
The guidewire runs into the inner lumen while the outer lumen is intended for contrast medium flow. The maximum diameter of the guide wire must not exceed 0.46 mm = 0.018 inches. Optionally, a guidewire measuring 0.014” = 0.36 mm can be used under medical criteria and in the cases where it is required by the lesion to be treated.
On the catheter’s distal part, just before the tip, there is the balloon (inflatable segment) that will dilate the artery upon inflating by means of infusion of contrast fluid inside it. The connector is Y-shaped and it has two entry ports:
– The straight port is intended for guidewire passage.
– The side port allows contrast medium flow to dilate the balloon
The balloon is coated with a homogeneous mixture of Paclitaxel, a derivative of Taxol, and a physiologically innocuous matrix, the excipient. The drug’s dose is 3 µg/mm2 of balloon surface and it is intended to avoid cellular proliferation, consequently decreasing re-intervention rate.
The drug is released from the balloon by means of a rapid inflation so that a high dose is released in a very short period of time. In order to assure a sufficient dosage of Paclitaxel onto the arterial wall, inflation process must last from 30 seconds to 1 minute. Dilation of the lesion can be optimized by using longer inflation times at operator’s discretion.
The balloon is designed to reach different diameters at different pressures, as predicted by the compliance curve included on the primary packaging.
Two radiopaque markers are located at each end of the balloon in order to mark its length and help the user to see the catheter while navigating inside the patient.
See IFU for further information. Available to download.
Indications
Dilatation of stenosis in iliac, femoral, iliofemoral, popliteal, infra-popliteal and renal arteries, as well as for the treatment of obstructive lesions of arteriovenous fistulas, whether original or artificial, with a reference diameter balloon from 2 to 8 mm and lengths from 20 to 200 mm. It is also indicated for stent post-dilation in the peripheral vascular system.
Features
- iVascular’s proprietary coating technology “Transfertech”. Nanotechnology drop dosage system that yields a multilayer thin coating.
- Improved coating mechanical resistance. Better adhesion to the balloon
- Homogeneous and precise Paclitaxel concentration of 3 µg/mm2 on the balloon for an effective treatment of the whole injury
- Microcrystalline coating for faster drug absorption rate. Rapid release of Paclitaxel with long-term efficacy.
- Segmented catheter design for excellent pushability and trackability. Balanced transition of forces. Anti-kinking performance.
- Pt-Ir radiopaque markers with high visibility
- Very short balloon deflation rates.
- Wide balloon size range (200mm lengths)
Specifications
- Catheter materials: Nylon/Pebax (the product does not contain latex components)
- Drug: 3 µg/mm2 Paclitaxel
- Excipient: Organic ester. Lipophilic, biocompatible and biodegradable.
- Balloon: Semi-compliant (10-15% from nominal pressure to RBP)
- Nominal Presure: 7 atm
- Rated Burst Pressure (RBP): 16 atm
- Average Burst Pressure (ABP): 20 atm
- Recommended guidewire: 0.018” (0.014” compatible)
- Crossing profile: from 0.035” up to 0.063”
- Tip profile: 0.019” maximum
- Introducer compatibility: 4F (d: 2 – 5 mm) |5F (d: 6 – 8 mm)
- Deflation time: < 8s
- Usable catheter lengths: 100 cm, 140 cm and 150 cm
- Available references

Clinical Support
Paclitaxel-eluting balloons have shown antiproliferative efficacy in the treatment and prevention of restenosis. Nevertheless, not all available devices are equally effective, which makes it interesting to compare results in a preclinical swine model (overexpansion 1.2 to 1.0). In this model, iVascular’s luminor DEB significantly reduced in-stent restenosis compared with the control balloons. Study published in the Spanish Journal of Cardiology (Rev Esp Cardiol. 2014;67:456-62 – Vol. 67 Num.06) and available for download on website.
Package Contents
- A paclitaxel-eluting dilatation balloon catheter OTW, covered by a protective shaft over the balloon and a protection stylet on the guidewire lumen. All the set is introduced in a circular dispenser to avoid damaging the catheter, and it is packed into a sterile bag.
- One card with the compliance curve showing the nominal inflation pressure and the recommended maximum pressure
- One leaflet with instructions for use